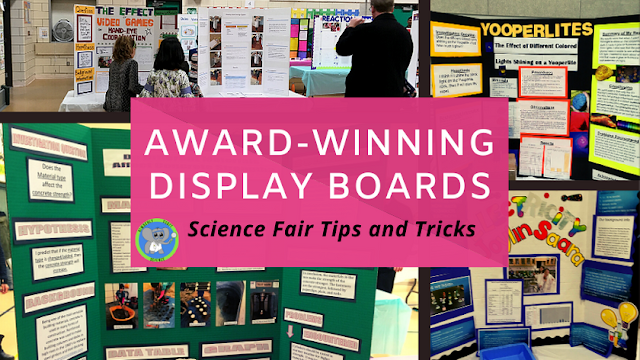
Reaction In A Bag Kid Version - Demonstrating Evidence Of A Chemical Change
Reaction in a bag is a wonderful classroom experiment for demonstrating the evidence of a chemical change. We have modified it to make it more kid-friendly for the classroom.
Reaction in a bag is a classic favorite. This lab allows students to break apart what is happening in the reaction. They have to use tables and data, and their evidence to guide them to the next step.
The premise of this lab is to discover what substances cause what parts of the reaction. They first observe the complete reaction with all 4 substances. The reaction produces heat, foaming, gas production, and a color change. Not all of the substances are a part of each stage. This is what makes the activity so interesting.
The premise of this lab is to discover what substances cause what parts of the reaction. They first observe the complete reaction with all 4 substances. The reaction produces heat, foaming, gas production, and a color change. Not all of the substances are a part of each stage. This is what makes the activity so interesting.
The students combine two substances at a time to discover their role in the reaction. It is low mess because all of it is done in a zip lock bag.
Performing the reaction in a bag also allows students to see the gas production and feel the bag get warm or cold. Some combinations of chemicals will do nothing. Others will be exothermic or endothermic reactions and may produce a color change. This will surely keep students engaged.
Check out this link for a ready-to-go activity, and then read the changes we made below to make it a little more budget and kid-friendly.
https://www.flinnsci.com/api/library/Download/a5d05c50dfd344e9af5972f81d0587cf
Changes to the Experiment.

1. Calcium Chloride
Check out this link for a ready-to-go activity, and then read the changes we made below to make it a little more budget and kid-friendly.
https://www.flinnsci.com/api/library/Download/a5d05c50dfd344e9af5972f81d0587cf
Changes to the Experiment.

1. Calcium Chloride
You do not need to order this from an expensive science supply catalog. Head over to your local superstar and purchase Damp Rid. You want to use the unscented, which is pure calcium chloride. It will save you money and you will have more than you need.
2. Phenyl Red
2. Phenyl Red
This is the biggest change I did. I did not have phenyl red, nor the budget to order some. I also find it harder to work with. I decided to use red cabbage juice. I boiled it in water until the color was mostly out of the cabbage and in the water.
Red cabbage produces a different change than the phenyl red. Both are indicators but produce different color changes. Red cabbage is an acid/base indicator. In the reaction with all 4 ingredients, it produced a purple color. Most likely because the substances were neutralizing. In the presence of a base it will turn more green. In the presence of an acid it will turn more pink.
When students began testing two items together, they quickly noticed that sodium bicarbonate and red cabbage juice turned green. Sodium bicarbonate is a base. This really interested them because it did not happen when all 4 substances were mixed. They were also surprised because the liquid got cool. When all 4 are mixed, the students feel the heat from calcium chloride and water reacting. This change will surprise them.
When the students mixed calcium chloride with red cabbage juice, they discovered it turned a bluish/green color. It did not seem to be quite as strong of a base as the baking soda, but it was close. In this reaction, a lot of heat was produced. Students immediately noticed more heat was produced than when all 4 substances were combined. This is most likely because the sodium bicarbonate and water are losing heat. It is exothermic.
Red cabbage produces a different change than the phenyl red. Both are indicators but produce different color changes. Red cabbage is an acid/base indicator. In the reaction with all 4 ingredients, it produced a purple color. Most likely because the substances were neutralizing. In the presence of a base it will turn more green. In the presence of an acid it will turn more pink.
When students began testing two items together, they quickly noticed that sodium bicarbonate and red cabbage juice turned green. Sodium bicarbonate is a base. This really interested them because it did not happen when all 4 substances were mixed. They were also surprised because the liquid got cool. When all 4 are mixed, the students feel the heat from calcium chloride and water reacting. This change will surprise them.
Each part will do different things and allow students to think like a scientist. If you have a group of 5th-8th graders, I strongly recommend this activity. It is memorable and really applies higher-order thinking skills. I recommend it for the start of a chemical reaction unit, where they will focus on the evidence of a chemical change.
After the activity, introduce the vocabulary and concepts. This reading passage complements the activity perfectly. Students will be able to relate it to the reaction and the evidence they observed.
SHOP THIS POST
LEARN MORE