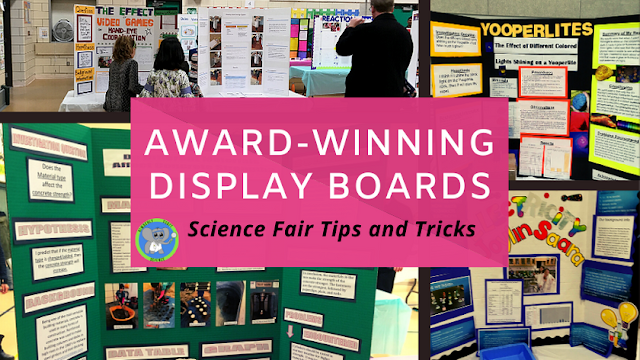
Demonstrating a Chemical Change for Halloween
During Halloween, students love to conduct exciting chemical reactions. Teachers want to engage the students but also impress the principal if they walk in. It's also crucial to ensure that our Halloween chemistry experiments meet the science standards.
Here are five chemistry experiments that will engage students and meet educational standards. They all focus on teaching the 5 signs of a chemical change.
1. Formation of a Gas
Effervescent Tablet Exploration
(Alka-Seltzer Tablets)
(Alka-Seltzer Tablets)
This reaction is perfect for teaching the formation of a gas. Think of it like the classic vinegar and baking soda reaction in a tablet. The tablet contains citric acid, which represents the acid. The tablet also contains sodium bicarbonate, which represents the base. When the tablet is placed in water, carbon dioxide gas is produced. To test that it is carbon dioxide gas, put a flame over the container. The carbon dioxide gas will smother the flame. If it was oxygen, the flame would glow brighter. You can add some dish soap to make it bubbly too.
2. Formation of a Precipitate
Falling Precipitate
This is a great example of the formation of a precipitate in a chemical reaction. Epsom Salt is hydrated magnesium sulfate. Washing soda is sodium carbonate. Sodium carbonate is used as a water softener in laundry detergent. Both are easy to find. When they are mixed together as solids, nothing happens. They need to be in an aqueous solution. When the solutions are combined, they are able to react to form the precipitate (solid), magnesium carbonate, and sodium carbonate (still in solution). The formation of the solid is an exciting surprise for students. Learn more in the video.
3. Unexpected Color Change
Red Cabbage Juice Indicator
Red cabbage juice is an acid/base indicator. This means it will change color in the presence of an acid or a base. It will turn red/pink in the presence of an acid and green in a base. The more acidic the pinker it is. The more basic, the more green. Students have a great time testing the red cabbage juice in different substances.
4. Temperature Change
Elephant Toothpaste
One of the best Halloween reactions involves erupting elephant toothpaste inside a pumpkin. This is an exothermic reaction, meaning it releases heat. It also gives off lots of bubbles of oxygen. Students will witness the heat and gas production firsthand. I've used this experiment for both class parties and in-class lessons on the scientific method.
5. Energy Production
Glow Stick Science
I find glow sticks fascinating. They are often used to keep children visible during trick-or-treating. Have you ever explained the science behind these glowing tubes to your students? A chemical reaction is responsible for producing the light; it is called chemiluminescence.
Have fun exploring evidence of a chemical change with students. Try all of them, or let the kids pick the one they want to try. It is sure to be memorable.
Shop this Post